We know that
• They made 20 sandwiches in total.
,
• Ben made three times as many as Ali made.
,
• Carla made twice as many as Ben.
Let's call Ben, Ali, and Carla B, A, and C.
The first statement would be expressed as
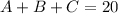
The second statement would be
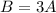
The third statement would be
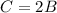
We have to find an equation with variable A only. Let's replace the second equation in the first one.
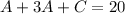
Then, we replace the second equation with the third one.
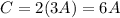
This last expression we include in the total sum to express it as A.
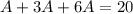
Then, we solve for A.
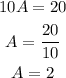
Therefore, Ali made 2 sandwiches.