We have to assume all the 20 g will react.
Now, the proportion on the equation is in moles, not in mass, so the first step is to convert the mass of TiCl₄ to number of moles of TiCl₄.
To do this, we will need the molar mass of TiCl₄, which we can calculate from theatomic weights of the atoms in int, Ti and Cl.
After we have the number o moles of TiCl₄, we can apply the proportion using the coefficients of TiCl₄ and the product we want, HCl. This will give the number of moles of HCl produced.
Lastly, we will need to convet this number of moles of HCl to mass of HCl, so we will also need the molar mass of HCl, which we can calculate from the atomic weights of H and Cl.
So, we will do the following:
1 - Calculate the molar masses of TiCl₄ and HCl
2 - Convert mass of TiCl₄ to number of moles of TiCl₄
3 - apply the proportion on the equation to convert from number of moles of TiCl₄ to number of moles of HCl
4 - Convert number of moles of HCl to mass of HCl.
1 - The molar mass is the sum of the atomic weights of each atom, so we will add 1 atomic weight of Ti and 4 atomic weights of Cl:
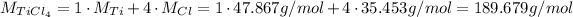
Now we do the same for HCl:
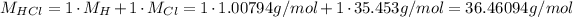
2 - The molar mass is the mass divided by the number of moles, so:
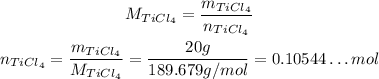
3 - Since the coefficient of TiCl₄ is 1 and the coefficient of HCl is 4, we have the following:
TiCl₄ --- HCl
1 --- 4
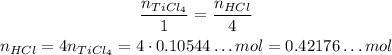
4 - Using the same equation for molar mass but now for HCl, we have:
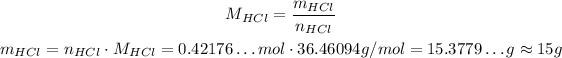
So, approximately 15g of HCl will be produced.