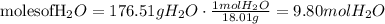1) Energy released from water
List known data
Volume: 171 mL H2O
Initial temperature: 82.4ºC
Final temperature: 0ºC
c=4.184 J/gºC
List unknown data
Energy released:
2) Set the equation
q=mcΔT
3) Change volume into mass
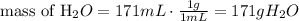
4) Replace known values
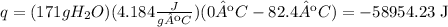
The total energy released -58954.23 J
5) The energy required to melt ice
List known data
q= -58954.23 J
ΔH(fus)= 334 J/g
List unknown data
m=
6) Set the equation
q=mΔH(fus)
7) Replace known values
+58954.23 J = m (334 J/g)
8) Solve the equation for m.
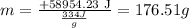
9) Find moles