Answer:
Explanations:
According to the Gay's Lussac's law, the pressure of a given mass of gas is directly proportional to the temperature provided that the volume is constant. Mathematically;
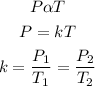
Given the following parameters
Initial pressure P1 = 11.5atm
Final pressure P2 = 3.3atm
Initial temperature T1 = 446.5K
Required
Final temperature T2
Substitute the given parameters into the formula
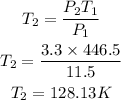
Hence the final temperature in kelvins is 128.13K