Chemistry =>Atomic Structure => Isotope
Isotopes are elements that have a different number of neutrons in their nucleus. Therefore, the atomic mass will be different between isotopes.
The atomic mass of lithium corresponds to the average atomic mass of its natural isotopes. To find the average we add the product of the mass of the isotope and its abundance, the formula for lithium will be in this case:
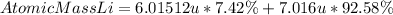
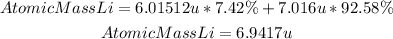
Answer: The atomic mass of lithium is 6.9417 u