The metal Strontium is symbolized by Sr, and since it is a metal its oxidation number is 0, so no charge.
In this reaction, the Sr will become 2+, so, since Cl is 1-, the proportion is 1 Sr to 2 Cl, so the unbalanced reaction is:
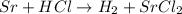
To balance it, we need to add a coefficient of 2 on HCl, so we have 2 H on both sides and 2 Cl on both sides.
The Sr is and continue balanced.
So, the balanced reaction is:
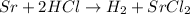
As we can see, the left part has Sr alone and H with Cl, while on the right side this changes, H is alone and Sr is with Cl. This means that Cl was displaced from H to Sr, so this is a single displacement reaction.