Answer:
a) pH= 13.3
b) pOH= 0.7
c) [H+]= 5.01x10^-14M
d) [OH-]= 0.2M
Step-by-step explanation:
The formula of sodium hydroxide is NaOH. From the dissociation of 1 mole of NaOH we obtain 1 mole of Na+ ion and 1 mole of OH- ion:
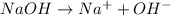
d) [OH-]
From the dissociation of 0.2M NaOH we will obtain a concentration of 0.2M Na+ and 0.2M OH-.
So, the concentration of OH- is 0.2M.
b) pOH
With the concentration of OH- and the formula of pOH we can calculate the pOH of the solution:
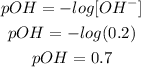
The pOH of the solution is 0.7.
a) pH
We can calculate the pH of the solution using the following formula, and replacing the value of pOH:
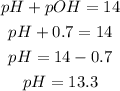
The pH of the solution is 13.3.
c) [H+]
Now with the pH formula we can calculate the concentration of H+:
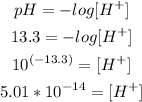
So, the concentration of H+ is 5.01x10^-14M.