Answer: There are 0.02 moles of solute in 10 mL of a 2.0 M Na2CO3 solution
Step-by-step explanation:
The question requires us to determine the amount of moles of solute that are in 10 mL of a 2.0 M Na2CO3 solution.
Considering that Na2CO3 is the solute in the solution given, we can use the concentration provided to calculate the amount of moles required.
From the concentration 2.0 M (or 2.0 mol/L), we know that there are 2.0 moles of Na2CO3 per liter of solution. Thus, we can write:
1 L = 1000 mL solution ----------------------- 2.0 mol Na2CO3
10 mL solutin ------------------------------------ x
Solving, for x, we'll have:
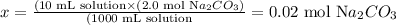
Therefore, there are 0.02 moles of solute in 10 mL of a 2.0 M Na2CO3 solution.