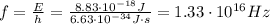In order to determine the frequency of the emmited electromagnetic energy, use the following formula:
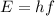
where:
E: energy of the photon = 8.83*10^(-18) J
h: Planck's constant = 6.63*10^(-34)J*s
f: frequency of the photon = ?
solve the previous equation for f, and replace the values of the other parameters, as follow: