1) Write the chemical equation.
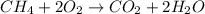
2) List the known and unknown quantities.
Sample: 2.7 g CH4.
Temperature: 380 K.
Pressure: 1.5 atm.
3) Convert grams of CH4 to moles of CH4.
The molar mass of CH4 is 16.0425 g/mol.
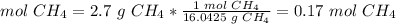
4) Moles of H2O produced from 0.17 mol CH4.
The molar ratio between CH4 and H2O is 1 mol CH4: 2 mol H2O.
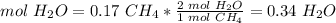
5) Volume of H2O produced in the reaction.
5.1- List the known and unknown quantities.
Moles: 0.34 mol H2O.
Temperature: 380 K.
Pressure: 1.5 atm.
Ideal gas constant: 0.082057 L * atm * K^(-1) * mol^(-1)
5.2- Set the equation.
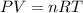
5.3- Plug in the known values and solve for V (liters).
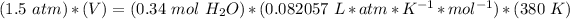
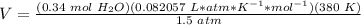
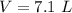
7.1 L H2O was produced from 2.7 g CH4 in the reaction.