By definition, the percent composition of an atom in a compound is its mass percentage in the formula.
That is, if we have 1 mol of NaHCO₃, we have also 1 mol of H (because there is only on H for each molecule).
So, we calculate the mass of this 1 mol of NaHCO₃ and the mass of 1 mol of H and calculate the percentage.
In equations, we want the following:
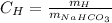
Since these are ratios, we doesn't matter if we talk about 1, 2 or any number of moles, but 1 mol is easier because the molecular and atomi masses are for 1 mol.
The molecular mass of NaHCO₃ is:
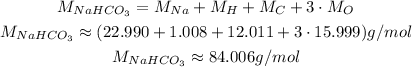
Which means that we have approximately 84.006 grams of NaHCO₃ in 1 mol of it.
The atomic mass of H is:
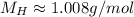
Which means that we have approximately 1.008 grams of H in 1 mol of it.
Now, we can take the percentage of mass of H:
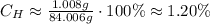
So, the percentage composition of H in NaHCO₃ is approximately 1.20%.