Answer: The pressure of the He is 2.97 atm
Step-by-step explanation:
According to Dalton's law, the total pressure is the sum of individual pressures.
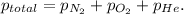
Given :
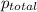 =total pressure of gases = 6.50 atm
=total pressure of gases = 6.50 atm
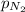 = partial pressure of Nitrogen = 1.23 atm
= partial pressure of Nitrogen = 1.23 atm
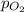 = partial pressure of oxygen = 2.3 atm
= partial pressure of oxygen = 2.3 atm
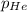 = partial pressure of Helium = ?
= partial pressure of Helium = ?
putting in the values we get:
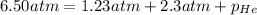
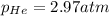
The pressure of the He is 2.97 atm