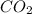Answer: a. 6.8 moles of water
b. 41.6 moles of carbon dioxide
Step-by-step explanation:
The balanced chemical equation for formation of water is:
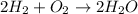
According to stoichiometry :
1 mole of
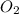 produces = 2 moles of
produces = 2 moles of
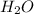
Thus 3.4 moles of
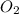 produce =
produce =
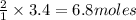 of
of
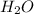
The balanced chemical equation for combustion of butane is:
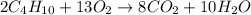
According to stoichiometry :
1 mole of butane produces = 8 moles of
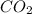
Thus 5.2 moles of butane produce =
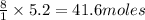 of
of