Answer:
The total change in enthalpy for the reaction is - 81533.6 J/mol
Step-by-step explanation:
Given the data in the question;
Reaction;
HCl + NaOH → NaCl + H₂O
Where initial temperature is 21.2 °C and final temperature is 28.0 °C. Ccal is 1234.28 J
Moles of NaOH = 50.mL × 1.00 M = 50.0 mmol = 0.0500 mol
Moles of HCl = 50.mL × 1.00 M = 50.0 mmol = 0.0500 mol
so, 0.0500 moles of H₂O produced
Volume of solution = 50.mL + 50.mL = 100.0 mL
Mass of solution m = volume × density = 100.0mL × 1.0 g/mL = 100 g
now ,
Heat energy of Solution q= (mass × specific heat capacity × temp Δ) + Cal
we know that; The specific heat of water(H₂O) is 4.18 J/g°C.
so we substitute
q_soln = (100g × 4.18 × ( 28.0 °C - 21.2 °C) ) + 1234.28
q_soln = 2842.4 + 1234.28
q_soln = 4076.68 J
Enthalpy change for the neutralization is ΔH
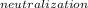
ΔH
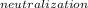 = -q_soln / mole of water produced
= -q_soln / mole of water produced
so we substitute
ΔH
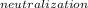 = -( 4076.68 J ) / 0.0500 mol
= -( 4076.68 J ) / 0.0500 mol
ΔH
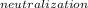 = - 81533.6 J/mol
= - 81533.6 J/mol
Therefore, the total change in enthalpy for the reaction is - 81533.6 J/mol