Answer: The chemical formula of the ionic compound formed by these elements when they react together is
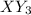 .
.
Step-by-step explanation:
For formation of a neutral ionic compound, the charges on cation and anion must be balanced. The cation is formed by loss of electrons by metals and anions are formed by gain of electrons by non metals.
Here element X is having an oxidation state of +3 as it can lose 3 electrons from its outermost orbit called as
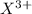 cation and element Y is having an oxidation state of -1 as it can gain one electron in its outermost orbit called as
cation and element Y is having an oxidation state of -1 as it can gain one electron in its outermost orbit called as
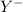 anion. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
anion. Thus they combine and their oxidation states are exchanged and written in simplest whole number ratios to give neutral
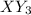 .
.