The potential energy of a single Ca−O ion pair that is just touching is approximately
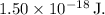
To calculate the potential energy (U) of a single Ca−O ion pair just touching, you can use Coulomb's law, which is given by the formula:
![\[ U = \frac{q_1 \cdot q_2}{{r}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/ztonawky0socofalqxhftoquavfgmiw9mk.png)
Where:
- k is Coulomb's constant (
 )
)
-
 are the charges of the ions (in Coulombs)
are the charges of the ions (in Coulombs)
- r is the distance between the centers of the ions (in meters)
Given ionic radii:
-
 (ionic radius of Ca in Ångströms)
(ionic radius of Ca in Ångströms)
-
 (ionic radius of O in Ångströms)
(ionic radius of O in Ångströms)
The distance (r) is the sum of the ionic radii when the ions are just touching:
![\[ r = r_{\text{Ca}} + r_{\text{O}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/rha50j53xvtshl57zn3u8k9ibxvw86jfkq.png)
Calculate the potential energy (U):
![\[ U = \frac{{k \cdot |q_{\text{Ca}} \cdot q_{\text{O}}|}}{{r}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/oi6dukxlpunh7k7825xv0fu8bgj4rlpvmf.png)
Now, considering that
 are the charges of Ca and O ions (which are typically +2 and -2 respectively), calculate U.
are the charges of Ca and O ions (which are typically +2 and -2 respectively), calculate U.
![\[ U = \frac{{(8.99 * 10^9 \, \text{Jm/C}^2) \cdot |(+2) \cdot (-2)|}}{{r_{\text{Ca}} + r_{\text{O}}}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/iw00fcmp12occbj3nenggelem2ptyfqt10.png)
![\[ U \approx \frac{{8.99 * 10^9 * 4}}{{1.76 + 0.66}} \, \text{J} \]](https://img.qammunity.org/2024/formulas/chemistry/college/25law2o4mzp4gywmkekqpjyhw8y0ticbqt.png)
![\[ U \approx 1.50 * 10^(-18) \, \text{J} \]](https://img.qammunity.org/2024/formulas/chemistry/college/6l3bxghqdvf2tcrcye8b9k6mp6jhv9r5tp.png)
The probable question may be:
From the ionic radii given in the figure (O= 0.66, Ca= 1.76), calculate the potential energy of a single Ca−O ion pair that is just touching.
e=1.60 x 10^-19C
k=8.99 x 10^9 Jm/C^2)