Answer:
Approximately
 , assuming that
, assuming that
 .
.
Step-by-step explanation:
This question is asking for the temperature change of the ball associated with a change in gravitational potential energy. While both the change in height
 and the specific heat
and the specific heat
 of the material are given, the mass
of the material are given, the mass
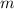 of the ball is not.
of the ball is not.
The change in the gravitational potential energy of this ball is:
 .
.
Under the assumptions, all that energy would have been converted into heat. In other words, the
 that has been lost would be equal to the heat energy
that has been lost would be equal to the heat energy
 that the ball has absorbed:
that the ball has absorbed:
 .
.
Note that there is a negative sign in front of
 in this expression because energy loss is denoted as a negative change, while energy gain is a positive change.
in this expression because energy loss is denoted as a negative change, while energy gain is a positive change.
Given the specific heat
 of the material, absorbing
of the material, absorbing
 of heat energy would raise the temperature of the material by:
of heat energy would raise the temperature of the material by:
 .
.
Note that the simplified expression for the temperature of the ball does not depend on the mass of the ball.
Substitute in
 ,
,
 (subtract initial value from the new value), and
(subtract initial value from the new value), and
 :
:
 .
.
(Note that
 .)
.)
In other words, the temperature of the ball would increase by approximately
 .
.