Final Answer:
The final temperature of the water after adding a 143°C platinum piece with a mass of 5.67g (c = 0.126) to 202g of 4.92°C water in a calorimeter that absorbs no heat is 22.43°C.
Step-by-step explanation:
When the hot platinum piece is added to the water in the calorimeter, heat transfer occurs until thermal equilibrium is reached. The heat lost by the platinum equals the heat gained by the water. The equation governing this heat transfer is given by the formula:
![\[ q = mc\Delta T \]](https://img.qammunity.org/2024/formulas/physics/high-school/ltij11nchnq47mlyrpofeis7ug352gt24k.png)
where
 is the heat transferred,
is the heat transferred,
 is the mass,
is the mass,
 is the specific heat, and
is the specific heat, and
 is the change in temperature. In this case, the heat lost by the platinum
is the change in temperature. In this case, the heat lost by the platinum
 is equal to the heat gained by the water
is equal to the heat gained by the water
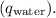
![\[ m_{\text{plat}}c_{\text{plat}}\Delta T_{\text{plat}} = m_{\text{water}}c_{\text{water}}\Delta T_{\text{water}} \]](https://img.qammunity.org/2024/formulas/physics/high-school/ddjuysuk5iycrmadgufn2x0jp2isxzwj58.png)
Given that the calorimeter absorbs no heat,
 Rearranging the equation and solving for the final temperature
Rearranging the equation and solving for the final temperature
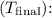
![\[ T_{\text{final}} = \frac{m_{\text{plat}}c_{\text{plat}}T_{\text{plat}} + m_{\text{water}}c_{\text{water}}T_{\text{water}}}{m_{\text{plat}}c_{\text{plat}} + m_{\text{water}}c_{\text{water}}} \]](https://img.qammunity.org/2024/formulas/physics/high-school/axk3nhqvno2tkhxko67d308zeucnz5v53g.png)
Substituting the known values into the equation, we find
 This final temperature represents the thermal equilibrium reached after the addition of the platinum piece to the water in the calorimeter.
This final temperature represents the thermal equilibrium reached after the addition of the platinum piece to the water in the calorimeter.