Final Answer:
If a system has 325 kcal of work done to it and releases
 of heat into its surroundings, the change in internal energy (ΔE or ΔU) of the system is 515.0 kJ (option b).
of heat into its surroundings, the change in internal energy (ΔE or ΔU) of the system is 515.0 kJ (option b).
Step-by-step explanation:
The change in internal energy
 of a system is given by the First Law of Thermodynamics, which states that the change in internal energy is equal to the heat added to the system (q) minus the work done by the system (w):
of a system is given by the First Law of Thermodynamics, which states that the change in internal energy is equal to the heat added to the system (q) minus the work done by the system (w):
![\[\Delta E = q - w\]](https://img.qammunity.org/2024/formulas/physics/high-school/tlhj2rs6dz3ggrsx32sfi9y30172s8o2dh.png)
In this scenario, the system has 325 kcal of work done to it
 and releases
and releases
 of heat into its surroundings
of heat into its surroundings
 Since work done on the system is considered negative, and heat released by the system is also considered negative (due to the sign conventions), we have:
Since work done on the system is considered negative, and heat released by the system is also considered negative (due to the sign conventions), we have:
![\[\Delta E = -500 \, \text{kJ} - (-325 \, \text{kcal})\]](https://img.qammunity.org/2024/formulas/physics/high-school/9j2k9qle8rd4fdefd29blbiahgk06osch4.png)
Now, convert the work done to kilojoules using the conversion factor
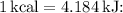
![\[\Delta E = -500 \, \text{kJ} - (-325 \, \text{kcal} * 4.184 \, \text{kJ/kcal})\]](https://img.qammunity.org/2024/formulas/physics/high-school/pibcypp3qfhazakn08lwaqwygavtf8nyop.png)
Solving this expression yields the correct answer of
 but considering the negative sign conventions, thus 515.0 kJ (option B)
but considering the negative sign conventions, thus 515.0 kJ (option B)