The total heat released during the process of condensing 58.0 g of water vapor at 100.0 °C to liquid water at 58.0 °C is approximately 131.244 kJ.
How to find total heat?
To calculate the total heat released during the process of condensing water vapor to liquid water, we can use the formula:
![\[ Q = m \cdot \Delta H \]](https://img.qammunity.org/2024/formulas/chemistry/college/ptmbef57xhtt9wqub1d4ysn0f0zdvg8w2m.png)
where:
Q = heat released,
m = mass of the substance (water in this case),
ΔH = enthalpy change for the phase transition.
For the condensation of water vapor to liquid water, the enthalpy change (ΔH) is the heat of vaporization (
 ).
).
Given:
Mass of water vapor (m) = 58.0 g
Heat of vaporization (
 ) for water at 100 °C =
) for water at 100 °C =
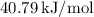
First, find the moles of water vapor using the molar mass of water (
 ):
):
![\[ \text{Molar mass of } H_2O = 18.015 \, \text{g/mol} \]](https://img.qammunity.org/2024/formulas/chemistry/college/lb06upiekui8jwk1ngp2rde0rvc6jqkvc0.png)
![\[ \text{Moles of } H_2O = \frac{\text{Mass}}{\text{Molar mass}} = \frac{58.0 \, \text{g}}{18.015 \, \text{g/mol}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/b23n5e64d490fd14andj9n86rwgv8nyag4.png)
Now, calculate the heat released:
![\[ Q = \text{Moles} * \Delta H_{\text{vap}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/kggoaufkn4si9in4jour0ma5i4zi7ar1tt.png)
![\[ Q = \left(\frac{58.0 \, \text{g}}{18.015 \, \text{g/mol}}\right) * (40.79 \, \text{kJ/mol}) \]](https://img.qammunity.org/2024/formulas/chemistry/college/zw6wjlt1os7cyl077lwno21g52g8z2ubmy.png)
![\[ Q = \text{Moles} * \Delta H_{\text{vap}} \]](https://img.qammunity.org/2024/formulas/chemistry/college/kggoaufkn4si9in4jour0ma5i4zi7ar1tt.png)
![\[ Q = (3.218 \, \text{mol}) * (40.79 \, \text{kJ/mol}) \]](https://img.qammunity.org/2024/formulas/chemistry/college/qo3is9umo8cfywk40c4y9xjftxaq3khhru.png)
![\[ Q \approx 131.244 \, \text{kJ} \]](https://img.qammunity.org/2024/formulas/chemistry/college/mbr1f4xdkfjr0h1er6e7ifiuqaxafmlvyl.png)
So, the total heat released during the process of condensing 58.0 g of water vapor at 100.0 °C to liquid water at 58.0 °C is approximately 131.244 kJ.
Complete question:
A 58.0 g sample of water vapor is condensing at 100.0 °C. Over time, all the water vapor is converted to liquid water at a temperature of 58 °C. Calculate the total heat released during this process.