Answer:
D) 1.20
Step-by-step explanation:
To find the final-to-initial volume ratio
 for an ideal gas undergoing an isochoric (constant volume) process, we can use Charles's Law.
for an ideal gas undergoing an isochoric (constant volume) process, we can use Charles's Law.
Charles's Law states that, at constant pressure, the volume of a given mass of gas is directly proportional to its absolute temperature.
The ratio of final to initial volume can be expressed as:
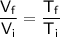
where:
 is the final volume,
is the final volume,
 is the initial volume,
is the initial volume,-
 is the final absolute temperature,
is the final absolute temperature, -
 is the initial absolute temperature.
is the initial absolute temperature.
Note: Temperatures must be in the absolute scale (Kelvin) for this equation.
Given that the initial temperature is
 and the final temperature is
and the final temperature is
 , we need to convert these temperatures to Kelvin:
, we need to convert these temperatures to Kelvin:
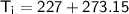
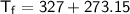
Now, substitute these values into the ratio equation:
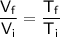
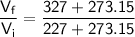
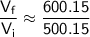
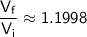
Now, round the result to a reasonable number of significant figures:
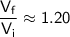
Therefore, the correct answer is option (D) 1.20.