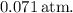The pressure of sulfur hexafluoride gas in the reaction vessel after the reaction is approximately
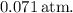
To calculate the pressure of sulfur hexafluoride gas in the reaction vessel, we can use the ideal gas law formula:
PV = nRT
Where:
- P is the pressure we want to find.
- V is the volume of the vessel, which is 35.0 L.
- n is the number of moles of the sulfur hexafluoride gas.
- R is the ideal gas constant,
- T is the temperature, which needs to be in Kelvin.
First, we convert the temperature from Celsius to Kelvin:
![\[ T (\text{K}) = T (\text{°C}) + 273.15 \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/lkll6f0c2js8b9xq5qb70aeonsiyakvnfj.png)
![\[ T = -13.0 + 273.15 \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/k83xio1to97m3xr5vacimgesm7lkrvoljk.png)
Next, we calculate the number of moles of sulfur hexafluoride (SF6). The molar mass of SF6 is the sum of the atomic masses of sulfur (32.07 g/mol) and six fluorine atoms (6 × 19.00 g/mol).
![\[ \text{Molar mass of SF6} = 32.07 + 6 * 19.00 \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/18q8d5itq6krtg1hzlp2fsu3ybqto41tk3.png)
Then we find the number of moles:
![\[ n = \frac{\text{mass}}{\text{molar mass}} \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/o2lvbkmlb755omga560zapme0t8ys48n44.png)
![\[ n = \frac{17.0 \, \text{g}}{\text{Molar mass of SF6}} \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/untj6npfs6xk4obadtuod56iausilikx08.png)
Finally, we rearrange the ideal gas law to solve for P :
![\[ P = (nRT)/(V) \]](https://img.qammunity.org/2024/formulas/chemistry/college/26ok1pzonmty7h1vp6rie559t6jzhnu33g.png)
Let's calculate these values step by step.
The pressure of sulfur hexafluoride gas in the reaction vessel after the reaction is approximately
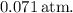
Here's the step-by-step calculation:
1. Temperature Conversion to Kelvin:
![\[ T = -13.0 + 273.15 = 260.15 \, \text{K} \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/cu9ihpn21gm417yrf69btkt7bbcja02b6c.png)
2. Calculate Molar Mass of SF6:
![\[ \text{Molar mass of SF6} = 32.07 + 6 * 19.00 = 146.07 \, \text{g/mol} \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/8aaa16k5a6fnq1fbk7di5jp6j12kw70y90.png)
3. Calculate Moles of SF6:
![\[ n = \frac{17.0 \, \text{g}}{146.07 \, \text{g/mol}} \]](https://img.qammunity.org/2024/formulas/chemistry/high-school/9fgebtoxn50avbms1mjsrhlwpe4jth0cr4.png)
4. Calculate Pressure using Ideal Gas Law:
![\[ P = (nRT)/(V) \]](https://img.qammunity.org/2024/formulas/chemistry/college/26ok1pzonmty7h1vp6rie559t6jzhnu33g.png)
Using the values, we find the pressure to be approximately