Answer:

Step-by-step explanation:
The number of grams of NaOH can be found by using the formula;
mass = number of moles × molar mass
Molar mass of NaOH = 40 g/mol
Since the number of particles was given the number of moles of the compound can be found by using the formula;
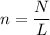
where n is the number of moles
N is the number of entities
L is the Avogadro's constant which is
6.02 × 10²³ entities
From the question;
N = 7.00 × 10²² molecules

The number of moles attained is then substituted into the formula for finding the mass of the compound.
