Answer:
2.9 moles of aluminum sulfate will be produced from the reaction of 5.8 moles of aluminum hydroxide and excess sulfuric acid.
Step-by-step explanation:
The balanced reaction is:
2 Al(OH)₃ + 3 H₂SO₄ → Al₂(SO₄)₃ + 6 H₂O
By reaction stoichiometry (that is, the relationship between the amount of reagents and products in a chemical reaction), the following amounts of each compound participate in the reaction:
- Al(OH)₃: 2 moles
- H₂SO₄: 3 moles
- Al₂(SO₄)₃: 1 mole
- H₂O: 6 moles
Then you can apply the following rule of three: if by reaction stoichiometry 2 moles of Al(OH)₃ produce 1 mole of Al₂(SO₄)₃, then 5.8 moles of Al(OH)₃ how many moles of Al₂(SO₄)₃ will they produce?
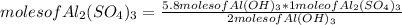
moles of Al₂(SO₄)₃= 2.9
2.9 moles of aluminum sulfate will be produced from the reaction of 5.8 moles of aluminum hydroxide and excess sulfuric acid.