The reaction has a 36.43% percent yield. This suggests that part of the reactants were not transformed into products and that the reaction did not proceed to its full potential.
What is moles ?
The mole, which is denoted by the symbol "mol," is the volume of a system that has the same number of atoms as there are in 0.012 kilo grammes of carbon 12.
The chemical equation for converting
 to
to
 is as follows:
is as follows:

3
 ⇒ 2
⇒ 2

From above we can conclude that the equation that 3 moles of
 react to create 2 moles of
react to create 2 moles of
 .
.
By applying the the mole ratio and molar mass of
 ,
,
the theoretical yield of O3 will be-
 mol mass = 3 x O mol mass = 3 x 16.00 g/mol = 48.00 g/mol.
mol mass = 3 x O mol mass = 3 x 16.00 g/mol = 48.00 g/mol.
o find the number of moles of O2, we can use the formula:
moles =

moles of
 =
=
 g/mol = 9.53 mol
g/mol = 9.53 mol
What is theoretical yield ?
The theoretical yield of
 from the balanced equation using the following formula:
from the balanced equation using the following formula:
Moles of
 =
=
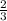 x moles of
x moles of

=
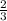 x 9.53 mol = 6.35 mol
x 9.53 mol = 6.35 mol
Mass of O3 = moles of
 x molar mass of
x molar mass of

= 6.35 mol x 48.00 g/mol = 304.80 g
= 304.80 g.
Percent yield = (
 ) x 100%
) x 100%
Given that the actual yield = 111 g
Percent yield = (
 ) x 100%
) x 100%
= 36.43%