Answer
47.73 g CuO
Step-by-step explanation
Given:
Chemical equation: 3CuO+ 2H3PO4 = Cu3(PO4)2 + 3H20
mass of orthophosphate acid (Cu3(PO4)2) = 39.2 g
Required: The mass of CuO
Solution:
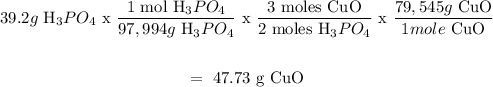
Second method:
Step 1: Find the moles of H3PO4
n = m/M where m is the mass and M is the molar mass of H3PO4
n = 39.2g/97.994g.mol^-1
n = 0.400 mol
Step 2: Use the stoichiometry to find the moles of CuO
The molar ratio between CuO and H3PO4 is 3:2
Therefore the moles of CuO = 0.400 mol x (3/2) = 0.600 mol
Step 3: Find the mass of CuO, now that we have moles
m = n x M m is the mass, n is the moles and M is the molar mass
m = 0.600 mol x 79,545 g/mol
m = 47.73 g