Mass Ag₂O needed = 107.416 g
Further explanation
Given
100 g Ag
Required
mass of Ag2O
Solution
Proust stated the Comparative Law that compounds are formed from elements with the same Mass Comparison so that the compound has a fixed composition of elements
mass of Ag in Ag2O :
= ((2 x Ar Ag)/molar mass Ag2O )x mass Ag2O
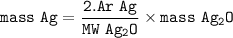
Ar Ag : 107,8682 g/mol
MW Ag₂O = 231,735 g/mol
Input the value :
mass Ag₂O=(mass Ag x MW Ag₂O) : (2 x Ar Ag)
mass Ag₂O = (100 g x 231,735 g/mol) : ( 2 x 107,8682 g/mol)
mass Ag₂O = 107.416 g