Answer: 7.77 g/ml
Step-by-step explanation:
Volume of cylinder with only water = 3.28 mL
Volume of cylinder with water and metal = 8.72 mL
Volume of metal = (Volume of cylinder with water and metal ) -(Volume of cylinder with only water)
=8.72-3.28
=5.44 ml
Mass of metal = 42.26 g
Formula of Density =
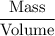
i.e. the density of the metal =
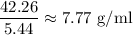
Hence, the density of metal = 7.77 g/ml