Answer:
Step-by-step explanation:
From the given information:
The compound in question here is
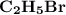
The molar mass of
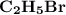 with 79 Br is;
with 79 Br is;
= (2 × 12) + (1 × 5) + 79
= 108 g/mol
The molar mass of
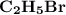 with 81 Br is:
with 81 Br is:
= (2 × 12) + (1 × 5) + 81
= 110 g/mol
Thus, the mass at which the molecular ions occurs are:
108 and 110.
The relative percentage of mass 108 occurred at 50.7%
The relative percentage of mass 110 occurred at 49.3%