Answer : The 3.01 moles of silver contains more number of atoms.
Explanation :
As we know that, 1 mole of substance contains
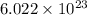 number of atoms.
number of atoms.
(a) The total number of atoms present in gold =

(b) The total number of atoms present in platinum =
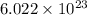
(c) As, 1 mole of silver contains
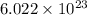 number of silver atoms.
number of silver atoms.
So, 3.01 mole of silver contains
 number of silver atoms.
number of silver atoms.
The total number of atoms present in silver =

Hence, from the above we conclude that, 3.01 moles of silver contains more number of atoms.