Answer : The correct option is (3) 500 K and 0.1 atm.
Explanation :
A real gas behaves ideally at high temperature and low pressure.
The ideal gas equation is,
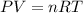
where,
P = pressure of gas
V = Volume of gas
R = Gas constant
T = temperature of gas
n = number of moles of gas
The ideal gas works properly when the inter-molecular interactions between the gas molecules and volume of gas molecule will be negligible. This is possible when pressure is low and temperature is high.
Therefore, the correct option is (3) 500 K and 0.1 atm.