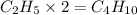Answer:
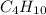
Step-by-step explanation:
Molecular formula is the chemical formula which depicts the actual number of atoms of each element present in the compound.
Empirical formula is the simplest chemical formula which depicts the whole number of atoms of each element present in the compound.
To calculate the molecular formula, we need to find the valency which is multiplied by each element to get the molecular formula.
Given empirical formula is
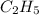 Empirical mass =
Empirical mass =
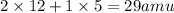
The equation used to calculate the valency is:
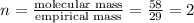
The molecular formula will be