Answer:
0.60 L
Step-by-step explanation:
Moles of
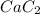 :-
:-
Mass = 1.53 g
Molar mass of
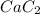 = 64.099 g/mol
= 64.099 g/mol
The formula for the calculation of moles is shown below:
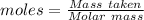
Thus,
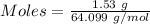
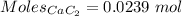
From the reaction shown below as:-
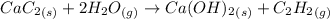
1 mole of
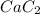 on reaction forms 1 mole of
on reaction forms 1 mole of
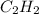
0.0239 mole of
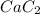 on reaction forms 0.0239 mole of
on reaction forms 0.0239 mole of
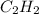
Mole of
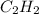 = 0.0239 mol
= 0.0239 mol
Vapor pressure of water = 21.07 torr
Total vapor pressure = 755 torr
Vapor pressure of
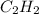 = Total vapor pressure - Vapor pressure of water = (755 - 21.07) torr = 733.93 torr
= Total vapor pressure - Vapor pressure of water = (755 - 21.07) torr = 733.93 torr
To calculate the amount of hydrogen gas collected, we use the equation given by ideal gas which follows:
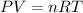
where,
P = pressure of the gas = 733.93 torr
V = Volume of the gas = ?
T = Temperature of the gas =
![25^oC=[23+273]K=296K](https://img.qammunity.org/2018/formulas/chemistry/college/s4x4fgn22qkdh8js274d4nosha2hcih4zh.png)
R = Gas constant =
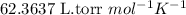
n = number of moles of
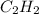 = 0.0239 mol
= 0.0239 mol
Putting values in above equation, we get:
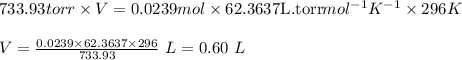
0.60 L is the volume of
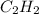 that is collected over water.
that is collected over water.