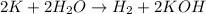Answer : The given balanced chemical reaction is,
Explanation :
Balanced chemical reaction : It is defined as the number of atoms of individual elements present on reactant side must be equal to the product side.
The given unbalanced chemical reaction is,
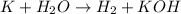
This chemical reaction is an unbalanced reaction because in this reaction, the number of atoms of hydrogen are not balanced while all the atoms are balanced.
In order to balanced the chemical reaction, the coefficient 2 is put before the
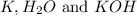 .
.
The given balanced chemical reaction is,