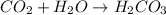Answer: The correct answer is
Step-by-step explanation:
Synthesis reaction is defined as the reaction in which two or more small substances combine together to form a single large compound. General equation representing this reaction follows:
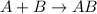
For the given reactions:
Reaction 1:
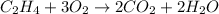
This reaction is considered as a combustion reaction because here, hydrocarbon is reacting with oxygen gas to produce carbon dioxide and water molecule.
Reaction 2:
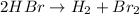
This reaction is considered as a decomposition reaction because here, one substance is forming two smaller substances.
Reaction 3:
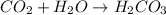
This reaction is considered as a synthesis reaction because here, two smaller substances are forming one single compound.
Reaction 4:
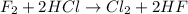
This reaction is considered as a single displacement reaction because here, a more reactive element is displacing a less reactive element.
Hence, the correct answer is