Answer:
True
Step-by-step explanation:
Valence electron:
No. of electrons present in the outermost shell of any atom is called valence electrons. Valence electrons determines the chemical properties of element.
Atomic no. of S = 16
Electronic configuration of S =
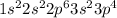
Energy shell (n = 3) is the outermost shell
Valence electrons = 6
Atomic no. of Cl = 17
Electronic configuration of S =
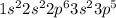
Energy shell (n = 3) is the outermost shell
Valence electrons = 7
So, sulfur has 6 valence electrons and Cl has 7 valence electrons.
The statement 'Sulfur has fewer valence electrons than chlorine.' is true.