Answer: Option (D) is the correct answer.
Step-by-step explanation:
When a chemical compound dissociates into ions in a solution then the resulting solution is known as electrolyte.
For example, HCl dissolved in water will act as an electrolyte.
And, when a chemical compound does not dissociate into ions when dissolved in a solvent then the resulting solution is known as a non-electrolyte.
For example,
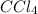 in water will act as a non-electrolyte.
in water will act as a non-electrolyte.
As a non-electrolyte does not contains ions therefore, they do not conduct electricity. Also, covalent compounds do not dissociate into ions when dissolved in water.
Thus, we can conclude that the statement non-electrolytes do not dissolve in water and can not conduct electricity, is true.