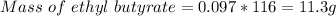Answer:
Amount of ethyl butyrate formed = 11.3 g
Step-by-step explanation:
The reaction between butanoic acid (C3H7COOH) and excess ethanol (C2H5OH) is:
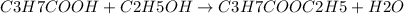
Since ethanol is the excess reagent, the formation of the product i.e. ethyl butyrate is influenced by the amount of butanoic acid present
Based on the reaction stoichiometry:
1 mole of butanoic acid produces 1 mole of ethyl butyrate
Mass of butanoic acid = 8.50 g
Molar mass of butanoic acid = 88 g/mol
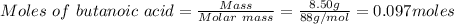
Moles of ethyl butyrate = 0.097
Molar mass of ethyl butyrate= 116 g/mol