Step-by-step explanation:
The sum of total charge of all the protons present inside the nucleus of an atom is known as the nuclear charge.
As the size of an atom increase there occurs a decrease in nuclear charge. This is because number of shells increases as the size of an atom increases. Therefore, force of attraction between the nucleus of the atom and its outermost electrons decreases.
It means electrons are shielded by the number of shells.
Due to this valence electrons can be easily lost by the atom in a chemical reaction.
Atomic number of magnesium is 12 and its electronic configuration is
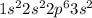 .
.
Hence, nuclear charge felt by the electrons present in 3s sub-shell will be the least.
Thus, we can conclude that electron is 3s subshell in magnesium is most shielded from nuclear charge.