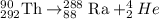Answer : The complete alpha decay reaction will be,
Explanation :
Alpha decay : In this process, alpha particles is emitted when a heavier nuclei decays into lighter nuclei. The alpha particle released has a charge of +2 units.
The general representation of alpha decay reaction is:
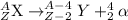
From the given reaction we conclude that,
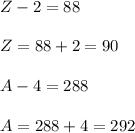
So, the element is thorium that has atomic number 90 and atomic mass 292.
The complete alpha decay reaction will be,