Answer: Four ionic compounds formed will be
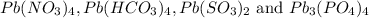
Step-by-step explanation:
Ionic compound is formed by the complete transfer of electrons from 1 atom to another atom. The cation is formed by the loss of electrons by metals and anions are formed by gain of electrons by non metals.
We are given:
One cation having formulas
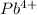
Four anions having formulas
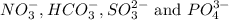
By criss-cross method, the oxidation state of the ions gets exchanged and they form the subscripts of the other ions. This results in the formation of a neutral compound.
The four ionic compounds are:
When lead ion and nitrate ions combine, it results in the formation of
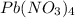 compound. This is named as lead (IV) nitrate.
compound. This is named as lead (IV) nitrate.
When lead ion and bicarbonate ions combine, it results in the formation of
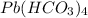 compound. This is named as lead (IV) hydrogen carbonate.
compound. This is named as lead (IV) hydrogen carbonate.
When lead ion and sulfite ions combine, it results in the formation of
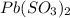 compound. This is named as lead (IV) sulfite.
compound. This is named as lead (IV) sulfite.
When lead ion and phosphate ions combine, it results in the formation of
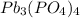 compound. This is named as lead (IV) phosphate.
compound. This is named as lead (IV) phosphate.