Answer: a. Because the carbon dioxide in the air mixes with it to form carbonic acid and d. Because sulfur dioxide released from burning petroleum mixes with it to form sulfuric acid.
Explanation:
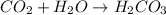
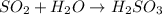 , which oxides in air to form
, which oxides in air to form
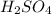
Thus carbonic acid
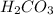 and sulphuric acid
and sulphuric acid
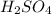 mixes with rain to form acid rain.
mixes with rain to form acid rain.
Nitrogen monoxide
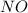 and not nitrous oxide
and not nitrous oxide
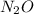 forms nitric acid.
forms nitric acid.