Answer: Option (a) is the correct answer.
Step-by-step explanation:
Molarity is defined as the number of moles present in a liter of solution.
Mathematically, Molarity =
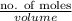
Since, it is given that molarity is 4.0 molar or 4.0 m/L and no. of moles is 12 moles.
Therefore, calculate volume as follows.
Molarity =
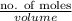
4.0 m/L =
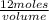
volume = 3 L
Hence, we can conclude that volume of the given solution is 3 liters.