Answer: The correct answer is option(D).
Step-by-step explanation:
According to thermodynamics, work done by the system is equal to the difference of the energy added to the system from change in internal energy of the system.
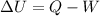 (First law of thermodynamics)
(First law of thermodynamics)
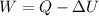
W = work done by the system
Q = Heat added to the system
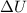 = Change heat energy of the system
= Change heat energy of the system
In an engine the change is internal energy difference can be considered as waste heat. This waste heat means that heat lost by the system.
The amount of work done by a heat engine equals the amount of thermal energy 'added to the engine minus the waste heat'.
Hence,the correct answer is option(D).