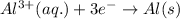Answer: The half reaction for reduction reaction is
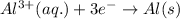
Step-by-step explanation:
Reduction reaction is defined as the reaction in which a substance gains electrons and the oxidation state of the substance is reduced.
Oxidation reaction is defined as the reaction in which a substance looses electrons and the oxidation state of the substance is increased.
For the given redox reaction:
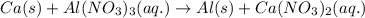
The half cell reactions for the above redox equation is:
Oxidation:
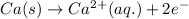
Reduction:
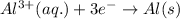
Hence, the half reaction for reduction reaction is