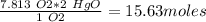Answer:
moles of HgO needed to produce 250.0 g of O2 = 15.63 moles
Step-by-step explanation:
The chemical reaction is:
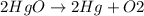
Amount of O2 produced = 250.0 g
Molar mass of HgO = 216.59 g/mol
Molar mass of O2 = 32.00 g/mol
Moles of O2 produced =
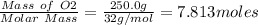
Based on the reaction stoichiometry:
2 moles of HgO produces 1 mole of O2
Therefore, moles of HgO needed to produce 7.813 moles of O2 is:
=