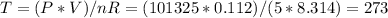Answer:
273 K
Step-by-step explanation:
We can assume an ideal gas and use the ideal gas law:
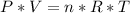
Where P is the pressure in Pascal, V is the volume in m^3, n is the number of moles, R is the universal gas constant in J/mol.K and T is the temperature in K.
We must convert the values to SI units.
1 L = 0.001 m^3
1 atm = 101325 Pa
Therefore the Pressure and Volume can be calculated as:
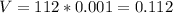
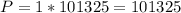
Therefore the Temperature in Kelvin can be determine from the ideal gas law: