Answer:
7.5 mol of oxygen are required
Step-by-step explanation:
Hi, as it says, this is a combustion reaction of benzene:
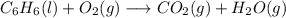
The first step is to balance the equation:
1 mol of
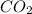 for each C in the benzene
for each C in the benzene
1 mol of
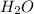 for each two H in the benzene
for each two H in the benzene
1 mol of
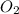 for each
for each
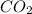 and 0.5 mol for each
and 0.5 mol for each
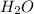
Balanced:

Now, following the reaction:
For 1 mol of benzene, 15/2 (7.5) mol of oxygen are required.