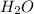Answer: 5.66 g of
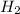 would be formed if 34 grams of carbon reacted with an unlimited amount of
would be formed if 34 grams of carbon reacted with an unlimited amount of
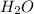
Step-by-step explanation:
According to avogadro's law, 1 mole of every substance occupies 22.4 L at STP and contains avogadro's number
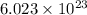 of particles.
of particles.
To calculate the moles, we use the equation:
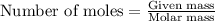
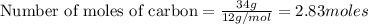
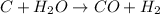
Carbon is the limiting reagent as it limits the formation of product and hydrogen is the excess reagent.
According to stoichiometry :
1 mole of carbon gives = 1 mole of
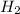
Thus 2.83 moles of carbon gives =
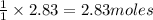 of
of
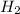
Mass of
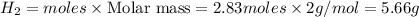
Thus 5.66 g of
 would be formed if 34 grams of carbon reacted with an unlimited amount of
would be formed if 34 grams of carbon reacted with an unlimited amount of