Answer: Option C is correct.
Explanation: Average kinetic energy is directly proportional to the absolute temperature. Higher the temperature means higher the kinetic energy.
Average kinetic energy is given by:
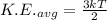
Where, k = Boltzman constant
T = Temperature
We are given different temperatures, so to compare they all should have the same units.
a) 298K
b) 267K
c) 27°C = 273+27 = 300K
d) 12°C = 273+12 = 285K
Looking at the temperature values, C part will have the highest average kinetic energy.